26 February 2026
The Trust, which runs the county’s acute hospitals, is involved in a study to explore whether shorter, higher-dose radiotherapy treatments can improve the quality of life for patients with advanced lung cancer.
Nine patients from The Shrewsbury and Telford Hospital NHS Trust (SaTH) have been recruited to the ‘Shortened High-dose Palliative Radiotherapy for Lung Cancer’ (SHiP-Rt) study.
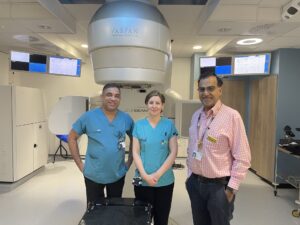 University Hospitals Coventry and Warwickshire (UHCW) NHS Trust is leading the study, which also involves participants from University Hospitals Birmingham NHS Foundation Trust and may open at the University Hospitals of North Midlands NHS Trust.
University Hospitals Coventry and Warwickshire (UHCW) NHS Trust is leading the study, which also involves participants from University Hospitals Birmingham NHS Foundation Trust and may open at the University Hospitals of North Midlands NHS Trust.
Currently, patients with advanced lung cancer typically receive 36 Gray (Gy) radiotherapy over 12 daily treatments, spanning about 16 days. The SHiP-Rt study is investigating whether delivering higher doses in six treatments given every other day could be a safe and feasible alternative using modern radiotherapy techniques.
The study aims to reduce the number of hospital visits for patients while saving radiotherapy slots for others, potentially improving efficiency and cutting costs for the NHS without increasing side effects.
Christopher Clarke, aged 88, from Telford, took part in the study when he was diagnosed with Stage 3b lung cancer.
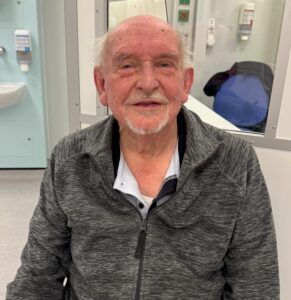 He underwent six sessions of radiotherapy and last month Mr Clarke was told by the specialist that the tumour had been reduced by two-thirds with no further treatment required at this stage.
He underwent six sessions of radiotherapy and last month Mr Clarke was told by the specialist that the tumour had been reduced by two-thirds with no further treatment required at this stage.
Mr Clarke said: “If I hadn’t taken part in this trial, radiotherapy would have been five days a week and I didn’t want my daughter and son-in-law having to take me to Royal Shrewsbury Hospital that much.”
“I think I am very lucky to receive this treatment. I can’t fault it – it was all very rapid. All very thorough and there didn’t seem much in the way of delays. It all rolled on very quickly.”
Patients are being recruited from across the West Midlands and it is hoped that results from this study will lead to a larger study involving more cancer treatment centres from around the UK.
Dr Anirban Chatterjee, Principal Investigator for the study, said: “I have worked closely with Dr Raj Srimali and it is my pleasure to state that SaTH has been one of the leading recruiters for SHiP RT trial. There has been excellent patient adherence and engagement.”
“Patients like the six-visit option instead of 12-13 visits for radiotherapy and no extra toxicity has been reported in the trial patients to date.”
The study at the end of November had already recruited 23 of 37 patients. Recruitment for the study ends on Tuesday 31 March and is due to end in March 2027.
Captions:
- Dr Chatterjee and research radiographers Emma Weaver and Sanal Jose
- Patient Christopher Clarke
